About CINRYZE
Not a real patient.
Untreated person with HAE
during an attack
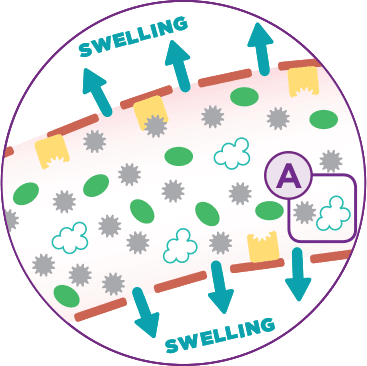

Missing or dysfunctional C1-INH causes increased levels of bradykinin
Person with HAE after
resolution of an attack


CINRYZE increases plasma levels of C1-INH to prevent the production of excess bradykinin
-

CINRYZE
-

Blood Vessel Wall
-

Bradykinin
-

Bradykinin B2 receptor
-

Missing/dysfunctional
C1 esterase inhibitor
-

Fluid Movement
-

Kallikrein
Want to learn more about an HAE treatment option that works differently from CINRYZE? Click here to learn more about one option to prevent HAE attacks in people 12 years of age and older.
Talk to your doctor about CINRYZE
Are you ready to see if preventive therapy might be right for you or your child?
Take the first step by talking with your doctor.
Download my discussion guide
You might also be interested in:
![CINRYZE® (C1 esterase inhibitor [human]) Logo](/dist/images/hdr-logo2.png)
